Stainless steel is well-known for resisting rust and corrosion. But even the best alloys can lose some of this protection after machining, welding, or polishing. These steps can leave behind tiny iron particles or heat marks that weaken the surface. This is where passivation becomes important. It’s a chemical process that restores and strengthens stainless steel’s natural protective layer.
Passivation does more than just clean the metal. It’s a precise treatment that directly affects how long stainless steel parts last in real conditions. From medical tools to marine parts, passivated surfaces stay clean longer, resist corrosion better, and meet stricter quality standards.

What Is Passivated Stainless Steel?
Passivation is a chemical process that improves the corrosion resistance of stainless steel. It removes free iron and helps form a chromium-rich oxide film on the surface. This process doesn’t add a coating or change the part’s size. Instead, it adjusts the surface so that oxygen can react with chromium in the alloy to form a thin, invisible oxide layer, usually 1–5 nanometers thick.
This layer acts as a barrier that blocks moisture and chemicals from reaching the base metal. Its best feature is that it can repair itself. When scratched, the exposed chromium reacts with oxygen to rebuild the protective film. That’s why well-passivated stainless steel can last for years without rusting.
How the Process Works?
Passivation has two main actions — cleaning and oxidation.
- The cleaning step removes oils, machining dust, and iron particles using controlled acid solutions.
- The oxidation step happens naturally when chromium reacts with oxygen to rebuild the protective film.
Why Stainless Steel Needs Passivation?
Even the highest-grade alloys lose protection during fabrication. Learn how manufacturing contamination weakens corrosion resistance and why chemical restoration is the key to durability.
Surface Contamination During Fabrication
Even top-quality stainless steel can get contaminated during fabrication. Processes like grinding, cutting, and welding can leave traces of carbon steel or create heat tints. These areas can later develop rust spots when exposed to air or humidity.
For example, a 304L stainless steel part may look perfect after welding, but if the weld isn’t cleaned and passivated, it can rust in as little as 48 hours during salt spray testing. This is not a metal defect — it’s a surface chemistry problem.
Restoring the Protective Chromium Layer
Passivation cleans the surface and restores the chromium-to-iron balance. When the chromium level is at least 1.5 times higher than iron, the surface becomes much more resistant to corrosion.
In ASTM A380 studies, passivated 316L stainless steel lasted more than 96 hours in salt spray tests without rust, while untreated samples showed discoloration within one day. This difference is crucial in food, medical, and pharmaceutical applications, where clean and durable surfaces are required.
Chemical Methods and Process Steps
Passivation relies on carefully balanced chemistry and process control. Here we break down the common acids, workflow, and parameters that ensure consistent surface protection.
Common Passivation Acids
Two main chemicals are used for stainless steel passivation — nitric acid and citric acid.
- Nitric Acid Passivation: This traditional method uses strong oxidizers to remove free iron and rebuild the oxide film. It works quickly and meets older standards like AMS 2700 Type II. However, it releases nitrogen oxide gases (NOx) and requires strong ventilation for safety.
- Citric Acid Passivation: This is a newer and eco-friendly method. It uses citric acid to bind and remove iron particles gently, without toxic fumes. Under ASTM A967 Type VI, citric acid can reduce waste disposal costs by up to 70% while maintaining the same corrosion resistance as nitric acid.
As more factories focus on safety and the environment, citric acid passivation is becoming the preferred choice.
Typical Process Workflow
- Pre-cleaning and Degreasing: Remove oil, grease, and particles using alkaline cleaners or ultrasonic tanks.
- Acid Immersion: Soak parts in nitric or citric acid for 20–30 minutes at 25–60°C, depending on the alloy.
- Rinsing and Neutralization: Rinse with deionized water and use a mild alkaline rinse to neutralize acid residue.
- Drying and Inspection: Dry the parts and check for a clean, even surface.
Each factor — acid strength, temperature, and soak time — must be controlled carefully. Short times can leave iron behind, and long times can dull the surface. Automated systems with pH and temperature controls keep the process within safe limits.
Key Process Parameters and Standards
Three factors affect the quality of passivation:
- Temperature: Higher temperatures make reactions faster but can dull the surface.
- Acid Concentration: Typically 10–25% nitric or 4–10% citric, depending on the grade.
- Time: Usually 15–45 minutes, enough to ensure a uniform protective layer.
To ensure quality, engineers use standards such as ASTM A967, ASTM A380, and AMS 2700. These define approved acids, dwell times, and testing methods.
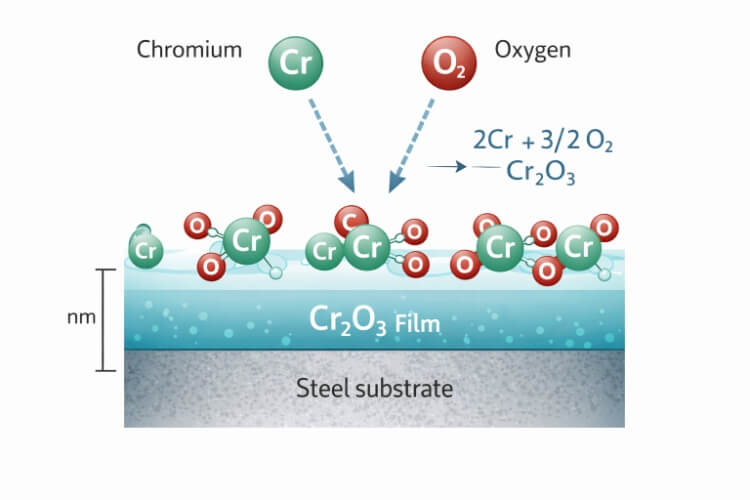
Microstructure and Surface Chemistry
Passivation transforms stainless steel at the microscopic level. This section explores how the chromium oxide layer forms, adapts, and maintains its self-healing corrosion resistance.
Formation of the Passive Film
At the core of stainless steel’s corrosion resistance is a very thin layer called the chromium oxide film (Cr₂O₃). When stainless steel undergoes passivation, chromium in the alloy reacts with oxygen to form this layer. Though it is only 1–5 nanometers thick, it’s extremely dense and stable. This invisible film protects the base metal from oxygen, moisture, and chloride attack.
Unlike the flaky rust (Fe₂O₃) that appears on carbon steel, chromium oxide is non-reactive and self-repairing. When scratched, new chromium near the surface reacts with oxygen to rebuild the layer almost instantly. This natural reaction keeps stainless steel clean, shiny, and corrosion-resistant for many years.
Influence of Alloy Composition
The effectiveness of passivation depends strongly on the alloy’s chemical makeup.
- Austenitic grades (304, 316): Contain high levels of chromium (18–20%) and nickel (8–10%), creating a smooth and uniform oxide film. Grade 316 also includes molybdenum (2–3%), which improves resistance to chloride corrosion — ideal for marine and chemical uses.
- Ferritic grades (430): Have lower nickel levels, making them more prone to oxidation. Careful control is needed during passivation to avoid etching.
- Martensitic grades (410, 420): Are harder and lower in chromium. They require extra cleaning and milder acids to prevent pitting.
Tests using SEM-EDS and XPS show that the chromium-to-iron ratio (Cr/Fe) on the surface determines corrosion resistance. A ratio above 1.5 gives strong protection, while untreated or poorly passivated surfaces often measure around 1.0.
Effect of Surface Finish
Surface texture also plays a big role in how well the oxide layer forms. Smooth, polished, or electropolished finishes provide fewer gaps for dirt or iron to collect, allowing a more even protective film. Rougher finishes, like as-machined or bead-blasted surfaces, trap microscopic particles and need longer or stronger acid treatment.
For food and pharmaceutical equipment, combining polishing with passivation can achieve a surface roughness (Ra) ≤ 0.8 µm, which meets hygiene and durability requirements.
Quality Control and Verification
Testing validates that passivation truly worked. Discover the analytical and visual inspection methods engineers use to confirm the protective film’s integrity and chemical balance.
Testing the Effectiveness of Passivation
Passivation quality must be verified through measurable testing, not just appearance. Common test methods include:
Water Break Test
A fully clean and passive surface lets water flow evenly over it. If the water forms drops, the surface still has contamination. This simple test is useful for quick checks.
Copper Sulfate Test (ASTM A967)
The surface is wiped or dipped in a copper sulfate solution for several minutes. If copper deposits form, it means free iron is still present. The results are visible within 10 minutes.
Salt Spray / Humidity Test (ASTM B117)
Samples are exposed to a salt mist for 24–96 hours. Properly passivated parts stay shiny, while untreated ones show rust within hours.
Advanced Analysis
For industries like aerospace and semiconductors, X-ray Photoelectron Spectroscopy (XPS) and Auger Electron Spectroscopy (AES) measure the oxide’s thickness and chemical composition at the nanometer level.
These tests confirm that the surface chemistry has shifted from iron-rich to chromium-rich — the key sign of successful passivation.
Visual and Analytical Inspection
Visual inspection is the first quality step. A properly passivated surface should look uniform, without stains, dull spots, or streaks. For precision parts, optical microscopes or digital analyzers can detect tiny contamination.
Measuring the Cr/Fe ratio adds another layer of proof. Research from the Journal of Materials Processing Technology shows that after proper passivation, the ratio on 316L stainless steel can increase to 2.0–2.5, nearly doubling corrosion resistance compared with untreated parts.
Documentation and Process Control
Consistency is just as vital as chemistry. Aerospace and medical industries follow AMS 2700 and ASTM A967 standards that record:
- Material type and batch number
- Acid type, concentration, and temperature
- Immersion time and rinse water quality
- Test results and inspector details
This traceability supports ISO 9001 and FDA audits, proving that each batch meets corrosion-resistance standards.

Key Industrial Applications
Passivated stainless steel is vital wherever corrosion or cleanliness directly affects performance:
- Food & Beverage: Piping, tanks, and fittings that must resist acids and cleaning agents.
- Medical & Pharmaceutical: Surgical tools and implants that need sterile, smooth surfaces.
- Aerospace & Automotive: Bolts, sensors, and lines exposed to heat, moisture, and vibration.
- Electronics & Semiconductors: Fixtures and housings where even minor rust can cause failure.
Proper passivation not only prevents corrosion but also ensures compliance and long-term reliability.
Environmental and Safety Considerations
As sustainability gains importance, manufacturers are rethinking traditional acid treatments. This section compares greener alternatives and how modern systems improve safety and efficiency.
Environmental Impact of Different Acids
The shift from nitric acid to citric acid has changed how manufacturers manage sustainability. Nitric acid reacts quickly but releases nitrogen oxides (NOx) — harmful gases that need costly ventilation and chemical neutralization. Poor handling can also lead to air or water contamination.
Citric acid offers a cleaner solution. It’s biodegradable, non-toxic, and works well at 4–10% concentration, compared to 20% or more for nitric acid. Tests under ASTM A967 Type VI show that citric acid can reduce hazardous waste by up to 70%, while matching or even improving corrosion resistance. Many ISO 14001 certified facilities now prefer citric acid passivation for its balance of performance, safety, and environmental responsibility.
Worker Safety and Handling Precautions
Even safe acids require careful handling. Operators should use chemical-resistant gloves, face shields, and aprons. Workstations must include fume hoods or closed tanks to reduce vapor exposure. Many modern plants now use automated passivation systems with enclosed chambers, temperature control, and pH monitoring. These systems protect workers and ensure process consistency within ±5%.
Used solutions are neutralized with sodium bicarbonate or calcium hydroxide before disposal. Continuous pH checks ensure compliance with EPA and REACH standards. By adding waste-neutralization and rinse-water recycling, manufacturers can reduce total discharge by about 40%, based on results from surface-treatment facilities.
Conclusion
Passivation is much more than a surface finish. It’s a key process that ensures stainless steel stays strong and corrosion-resistant. By removing free iron and rebuilding a stable chromium-oxide film, it turns stainless steel into a truly reliable material for long-term use.
When done under standards like ASTM A967, ASTM A380, or AMS 2700, passivation improves salt-spray resistance, surface cleanliness, and part lifespan. For manufacturers, that means fewer failures, less maintenance, and stronger product trust.
Ready to enhance your stainless steel components? See how professional passivation support can improve surface performance, extend lifespan, and ensure every part meets specification. Reach out today to discuss your project with our technical team.
FAQs
What is the difference between cleaning and passivation?
Cleaning removes oils and dirt. Passivation removes embedded iron and rebuilds the chromium oxide layer that prevents corrosion.
How often should parts be re-passivated?
Usually only once, right after fabrication. Parts that go through abrasive cleaning or welding repair may need re-treatment.
Can all grades of stainless steel be passivated?
Yes. Austenitic grades (304, 316) respond best, while ferritic and martensitic types need milder acid and tighter control.
Is citric acid passivation as effective as nitric acid?
Yes. ASTM tests confirm equal or better corrosion protection with fewer safety and environmental risks.
How can I verify if passivation was successful?
Basic field tests like the water-break or copper-sulfate test show surface cleanliness. For high-precision parts, lab tests such as XPS or salt-spray analysis confirm oxide quality and Cr/Fe ratio.