In the world of high-precision metal fabrication, raw steel rarely arrives ready for the assembly line. When steel is processed at the mill—rolled red-hot at temperatures exceeding 1,000°F—it reacts violently with oxygen. As it cools, this reaction forms a hard, bluish-black crust known as mill scale.
While this scale is a natural byproduct, it is the silent killer of finish quality. If you paint over it, the coating will eventually peel as the scale detaches. If you weld through it, the impurities will create porosity and weak joints.
This is where Steel Pickling becomes non-negotiable. It is the critical “bridge” process that transforms raw, hot-rolled stock into a chemically clean substrate ready for automotive, aerospace, and appliance applications.

What is Steel Pickling?
Steel pickling is a surface treatment process that cleans metal using a chemical solution called “pickle liquor.” This solution removes rust, scale, and other unwanted materials from metals like steel, copper, and aluminum alloys.
The metal is placed in a strong acid bath that dissolves the oxide layer but does not harm the base metal underneath. After pickling, the surface is smooth, clean, and meets the SSPC-SP 8 (Pickling) standard for industrial cleanliness.
What is the Purpose of Pickling?
Pickling isn’t just about aesthetics; it is about structural integrity and chemical adhesion. To understand why this step is critical, we must first understand the “enemy” it removes.
Removing Impurities: Explain the target contaminants
Mill scale is not just dirt; it is a complex, layered structure of iron oxides:
- Wüstite (FeO): The layer closest to the metal.
- Magnetite (Fe₃O₄): The intermediate layer.
- Hematite (Fe₂O₃): The outer, brittle layer.
The Problem: Mill scale is electrically insulating and physically brittle. Unlike the ductile steel beneath it, scale will crack under stress. If you attempt to cold-roll steel without pickling it first, this hard scale will be pressed into the softer surface, permanently damaging the steel and ruining expensive rolling dies.

The Goal: “White Metal” Activation
For downstream processes, “clean” isn’t enough—the surface must be chemically active.
- For Coatings & Painting: Paints and galvanizing layers rely on a specific surface profile (anchor pattern) to grip the metal. Pickling removes the passive oxide layer, exposing the reactive “white metal” that bonds aggressively with primers and zinc coatings.
- For Precision Welding: Welding on dirty steel introduces oxygen and impurities into the weld pool. Pickling ensures a contaminant-free zone, which is essential for robotic welding lines where consistency is paramount.
Pro Tip:
Many fabrication failures—such as powder coating delamination after six months—can be traced back to improper pickling or “under-pickling” where residual scale was left behind. At TZR, we view surface preparation as the foundation of product longevity.
How the Pickling Process Works?
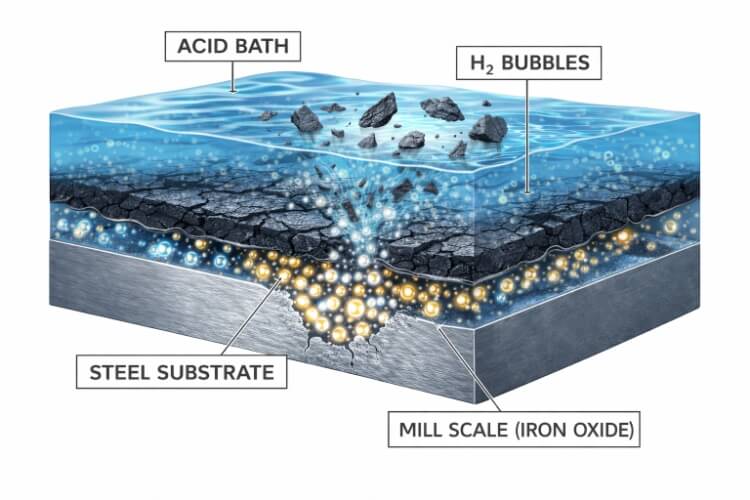
While it might look like a simple dunk tank, pickling is a violent, carefully controlled chemical assault on impurities. To achieve a perfectly clean surface without dissolving the valuable steel underneath, manufacturers rely on a specific mechanism: the “Blast Off” Effect.
The Chemical Mechanism
Many assume the acid simply eats the scale layer from the top down. In reality, the process is far more dynamic.
Mill scale is naturally porous and full of microscopic cracks. The pickling acid (the “pickle liquor”) seeps through these fissures and attacks the intermediate oxide layer (FeO) right at the interface of the base metal.
As the acid reacts with the iron, it generates hydrogen gas. These tiny hydrogen bubbles form underneath the scale layer, building up immense pressure. Like millions of micro-explosives, this pressure literally blasts the scale off the surface of the metal, causing it to flake away into the solution. Simultaneously, the acid dissolves the remaining iron oxides, leaving behind a pristine, chemically active steel surface.
Expert Note:
The speed of this reaction is critical. If the steel stays in the bath too long, “over-pickling” occurs. The acid begins to attack the base metal itself, causing severe pitting and material loss. This is why precise dwell times are non-negotiable for quality control.
The Step-by-Step Workflow
A successful pickling line involves a rigorous sequence of steps designed to ensure the metal is clean, processed, and protected.
Step 1: Degreasing and Cleaning
Before the steel ever touches the acid, it must be free of organic contaminants. Oils, greases, and drawing lubricants act as a barrier, preventing the acid from reaching the surface. A hot alkaline cleaner or solvent degreaser is used first; otherwise, the acid will simply bead up on the oily spots, leaving patches of unpickled scale.
Step 2: The Acid Bath (Pickling)
This is the core stage. The metal is submerged or sprayed with the acid solution. Modern continuous lines use high-pressure spray nozzles to agitate the acid, ensuring fresh solution constantly hits the surface, which speeds up the reaction significantly compared to static dipping.
Step 3: Rinsing (Cascade Rinse)
Once the scale is removed, the chemical reaction must be stopped immediately. The steel goes through a rigorous water rinse—often a multi-stage cascade—to wash away all traces of acid and dissolved metal salts. Any residue left behind will cause severe corrosion later.
Step 4: Neutralization
Rinsing with water isn’t always enough to neutralize the pH of the steel surface. A weak alkaline bath (often using ammonia or lime) is applied to ensure the surface is completely passive and no acidic ions remain in the metal’s pores.
Step 5: Oiling or Sealing (Critical for “Flash Rust”)
This final step is the difference between a usable product and scrap. Freshly pickled steel is highly reactive—it is essentially “naked” iron. Without protection, it will react with oxygen in the air almost instantly, forming a layer of yellow “flash rust.” To prevent this, the steel is immediately coated with a thin layer of oil, a rust inhibitor, or a water-soluble wax to seal the surface for storage or shipping.
Application Methods: Immersion vs. Spray
The physical method of applying the acid depends largely on the shape of the product:
- Batch Pickling (Immersion): Used for pipes, fabricated parts, or bundles of rods. The items are placed in acid-resistant crates or on racks and dipped into large vats of acid. This ensures the liquid reaches inside tubes and complex geometries.
- Continuous Pickling (Spray or Drag-Through): Used primarily for steel coils. The steel strip is unrolled and pulled continuously through a series of tanks at high speeds (up to 1,000 feet per minute).
Types of Pickling Acids: HCl vs. H2SO4
While the goal of pickling is uniform—removing oxides—the chemical agent used to achieve it fundamentally alters the process speed, cost, and, most importantly, the surface finish of your product.
In the carbon steel industry, two acids dominate: Hydrochloric Acid (HCl) and Sulfuric Acid (H₂SO₄). The choice between them is often a trade-off between operational cost and surface quality.
Hydrochloric Acid (HCl): The Precision Standard
Since the mid-20th century, HCl has become the overwhelming favorite for continuous sheet pickling lines, particularly for automotive and appliance grade steels.
- The “Bright” Finish: HCl attacks the scale layers (FeO, Fe₂O₃, Fe₃O₄) aggressively but is relatively gentle on the base metal. This results in a brighter, whiter, and cleaner surface with less “smut” (insoluble carbon residue) compared to sulfuric acid.
- Energy Efficiency: One of its biggest advantages is temperature. HCl works effectively at ambient temperatures or with minimal heating (typically 160°F – 180°F), significantly reducing the line’s energy consumption.
- The Trade-off: The major drawback is volatility. HCl fumes easily, requiring expensive ventilation and scrubbing systems to protect workers and equipment. It is also more expensive to purchase per ton than sulfuric acid.
Best For: High-quality sheet metal, automotive panels, and parts requiring plating or painting.
Sulfuric Acid (H₂SO₄): The Economic Workhorse
Before the 1960s, Sulfuric Acid was the industry standard. It remains popular today for batch pickling rods, wires, and structural shapes where cost recovery is a priority.
- Renewability: Sulfuric acid is cheaper to buy and easier to renew. As the acid becomes saturated with iron, the ferrous sulfate can be crystallized out, allowing the acid to be regenerated and reused almost indefinitely.
- The Heat Requirement: To work effectively, Sulfuric Acid must be heated to 180°F -225°F (82°C – 107°C). While the chemical is cheap, the energy bill to keep thousands of gallons of acid near boiling point is substantial.
- The Finish Risk: The resulting surface is often darker and may have more residual smut. It also poses a higher risk of “over-pickling”—if the line stops, the hot acid will continue to attack the base steel aggressively, pitting the surface.
Best For: Rebar, tubing, structural pipe, and low-carbon wire where surface aesthetics are secondary to cost.
Specialty Acids for Tougher Alloys
Standard acids often fail when dealing with high-alloy materials.
- High Carbon Steel (>0.6% Carbon): As carbon content rises, the steel becomes more resistant to standard pickling. Phosphoric Acid, Nitric Acid, or Hydrofluoric Acid may be added to the mix to aid in scale removal.
- Stainless Steel: The chromium oxide layer on stainless steel is incredibly tough. It typically requires a fierce combination of Nitric and Hydrofluoric acids (often called “mixed acid”) to clean effectively and restore passivity.
Quick Selection Guide: Which Acid Do You Need?
| Feature | Hydrochloric Acid (HCl) | Sulfuric Acid (H2SO4) |
|---|---|---|
| Primary Application | Continuous Sheet/Strip (Auto/Appliance) | Batch Pickling / Rod & Wire |
| Operating Temp | Lower (<180°F) – Energy Saver | Higher (180°F – 225°F) – Energy Heavy |
| Surface Result | Bright, White, Clean | Darker, potential smut |
| Reaction Speed | Faster (2–3x speed of H2SO4) | Slower |
| Cost Profile | High Chemical Cost / Low Energy | Low Chemical Cost / High Energy |
Pro Tip:
If your project involves subsequent electroplating or high-gloss powder coating, specifying HCl-pickled steel is often the safer bet. The brighter surface profile ensures better adhesion and fewer cosmetic defects than sulfuric-pickled alternatives.
Critical Challenges and Solutions
While pickling is standard practice, it is not without peril. For engineers and procurement managers, understanding the risks of Hydrogen Embrittlement and Hazardous Waste is just as important as the surface finish itself. Ignorance here can lead to catastrophic part failures or supply chain liabilities.
Hydrogen Embrittlement
One of the most dangerous side effects of pickling—and the one most often overlooked in basic guides—is Hydrogen Embrittlement (HE).
The Mechanism:
During the aggressive acid reaction, hydrogen gas is generated. While most bubbles away, tiny atomic hydrogen is small enough to diffuse directly into the steel’s crystal lattice. Once inside, these atoms migrate to areas of high stress (like grain boundaries or welds) and recombine into molecular hydrogen (H2).
This internal pressure builds up like a balloon inflating inside a rock. It creates immense internal stress that drastically reduces the metal’s ductility.
The “Delayed Failure” Nightmare:
The terrifying aspect of HE is that it causes delayed fracture. A bolt or bracket might pass QC inspection perfectly, only to snap suddenly under load weeks after installation. There is often no warning—no bending, no stretching—just a sudden, brittle snap.
The Solution: ASTM B850 Baking
- Inhibitors: Modern pickling liquors use chemical inhibitors to form a protective film on the bare metal, slowing down hydrogen absorption.
- De-embrittlement Baking: For high-strength steels (typically those harder than 30-32 HRC), parts must undergo a “baking” process immediately after pickling.
- Standard: According to ASTM B850, parts should be baked at approx. 375°F -400°F (190°C – 205°C) for at least 3 hours (often up to 24 hours depending on tensile strength) to force the trapped hydrogen out before it causes permanent damage.
Expert Note:
If you are sourcing high-tensile fasteners or springs that require pickling, always request a certificate of baking. Skipping this step is the #1 cause of unexplained fastener failure in the field.
Management of “Pickling Sludge”
The byproduct of the pickling process is not just dirty water; it is a hazardous toxic waste known as Spent Pickle Liquor (SPL).
The Environmental Cost:
As the acid works, it becomes saturated with dissolved iron salts (ferrous chloride or sulfate) and heavy metals (chrome, nickel, lead) stripped from the alloy. Once the acid concentration drops too low to be effective, this “sludge” must be treated.
Disposal & Regeneration:
- Neutralization: The most common treatment involves adding lime (calcium hydroxide) to the spent acid. This neutralizes the pH and causes the heavy metals to precipitate out as a solid sludge, which is then sent to hazardous landfills.
- Acid Regeneration Plants (ARP): Advanced facilities—particularly those using Hydrochloric Acid—use roasting processes to recover the acid gas for reuse. This creates a closed-loop system and produces iron oxide powder, a valuable byproduct used in the manufacturing of ferrite magnets and pigments.
Economic Considerations
Strict environmental regulations regarding SPL disposal have driven up the cost of pickling services in Western markets. When comparing quotes, be wary of suppliers with “too good to be true” pricing; they may be cutting corners on waste management or using exhausted acid baths that result in poor surface quality.
Alternatives to Chemical Pickling: When to Ditch the Acid
While acid pickling is the undisputed king of high-volume sheet metal production, strict environmental regulations and the specific risk of hydrogen embrittlement have driven the development of alternative methods. For certain applications, these “dry” techniques may be superior.
Mechanical Cleaning (SPS / Abrasive Blasting)
For structural steel projects where chemical handling is impossible or where a specific surface texture is required, mechanical cleaning—often governed by ISO 8501-1 standards—is the go-to choice.
- The Process: Steel is blasted with abrasive media (sand, grit, or steel shot) or aggressively wire-brushed.
- Pros: It eliminates hazardous acid waste and completely avoids Hydrogen Embrittlement risks. It is ideal for heavy rust removal on beams and plates.
- Cons: It leaves a rough, matte finish compared to the smooth, bright surface of acid pickling. It also struggles to clean internal surfaces of tubing or complex shapes where blast media cannot reach.
Flame Cleaning
- The Process: An oxy-acetylene flame is passed over the steel surface. The rapid thermal expansion causes the brittle mill scale to flake off, while the base metal expands at a different rate.
- Pros: Useful for localized cleaning or preparing existing structures for repainting where chemical baths are impractical.
- Cons: High energy cost and a high potential to warp thin materials due to heat input.
Laser Cleaning (The Future of “Green” Surface Prep)
- The Process: High-intensity pulsed lasers ablate (vaporize) the oxide layer and rust instantly.
- Pros: This is the most environmentally friendly option available. It produces no chemical waste, requires no consumables, and offers precise control over how much material is removed without damaging the substrate.
- Cons: The initial equipment cost is extremely high, and cleaning speeds for large surface areas are currently much slower than a continuous acid pickling line.
Conclusion
Steel pickling is far more than just a “cleaning” step; it is a fundamental metallurgical process that defines the DNA of your final product. Whether you are welding, painting, or cold-rolling, the success of your operation depends on the microscopic purity of the metal surface.
For buyers and engineers, specifying “Pickled & Oiled” is not just a line item on a PO—it is an assurance that your steel is free from hidden defects and ready for high-performance manufacturing.
At TZR, we don’t just cut and bend metal; we engineer quality from the substrate up. Our team leverages over a decade of sheet metal fabrication experience to ensure that every material we use meets rigorous surface quality standards.
Whether you need perfectly pickled sheet metal for a flawless powder-coated finish or robust structural components free from hydrogen risks, we have the expertise to guide your choice. Ready to secure your supply chain? Contact TZR Today for a consultation on your next project.




