Stainless steel is inherently corrosion-resistant. Plating it is a calculated engineering decision to alter surface mechanics, not a rust-prevention measure. We specify nickel plating to drastically improve wear resistance, increase surface hardness, eliminate thread galling, and enable reliable soldering or brazing during final assembly.
Nickel plating stainless steel significantly boosts corrosion resistance, surface hardness, and solderability. To ensure superior adhesion, the metal’s passive oxide layer must be removed via a Woods Nickel Strike before applying electroplated or electroless nickel. This specialized activation prevents peeling and ensures a durable, wear-resistant finish for industrial components.
This guide focuses strictly on the practical, shop-floor realities of plating stainless steel. We break down process selection, adhesion control, tolerance planning, and the physical cost trade-offs that dictate large-scale manufacturing.

Why Stainless Steel Is Difficult to Plate?
Stainless steel resists plating due to its stable surface layer. It blocks adhesion and requires specific activation steps before coating.
The Passive Oxide Layer
The defining characteristic of stainless steel is its chromium-rich passive oxide layer. This film forms instantly upon exposure to oxygen, protecting the substrate but acting as a physical barrier to electroplating.
Depositing nickel directly over this passive layer results in “false adhesion.” The plating may visually pass inspection in the tank, but it will inevitably blister or peel the moment it faces mechanical stress during a standard bend test or thermal cycling.
Wood’s Nickel Strike
You cannot bypass the passive layer; it must be chemically stripped while simultaneously preventing it from reforming. The industrial standard for this is a Wood’s Nickel Strike.
- The Chemistry: This process uses a highly acidic bath—a precise ratio of nickel chloride and hydrochloric acid—run at a very high current density.
- The Mechanism: It aggressively etches away the oxide layer and instantly flash-deposits a micro-thin, highly active layer of nickel, creating the necessary metallurgical bond.
Surface Activation Limits
Activation requires aggressive, targeted chemical management. Different grades of stainless steel react differently on the line.
High-alloy grades like 316, or parts that have undergone work hardening during sheet metal stamping, require much more rigorous cathodic activation compared to a standard 304 sheet. Failing to fully activate the specific surface geometry guarantees the initial strike will fail.
Strict Transfer Times
In strict production environments, adhesion failures frequently happen between the tanks, not inside them. Once the Wood’s strike is applied, the surface is highly reactive.
If the transfer time between the strike tank and the primary nickel bath is too slow—often needing to be kept well under 30 seconds—the part will dry or re-passivate in ambient air, causing downstream delamination.
Process Selection: Electroless Nickel Plating vs. Electrolytic Nickel
Different plating methods produce very different results in real parts. Each process changes cost, coverage quality, and functional behavior.
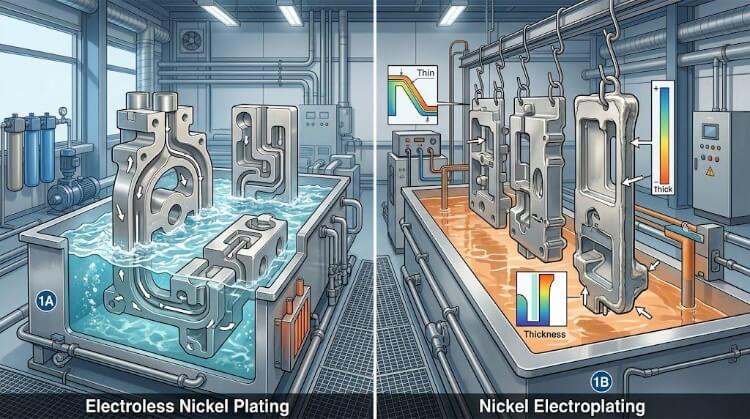
Electroless Nickel Plating (ENP)
ENP is an autocatalytic chemical reduction process that requires no external electrical current. It co-deposits nickel and phosphorus, making it highly predictable for precision manufacturing.
- Controlled Hardness: By controlling the phosphorus content (low, medium, or high), engineers can accurately dial in specific hardness levels and wear resistance.
- Absolute Uniformity: Deposition rate is driven entirely by bath temperature and chemistry. If a drawing specifies a 15-micron coating, the part receives exactly 15 microns uniformly across all surfaces.
- Complex Geometries: ENP is the only viable option for CNC machined parts with internal threads, deep recesses, or intersecting cross-holes. It ensures internal surfaces receive the exact same protection as the exterior.
Electrolytic Nickel
Electrolytic nickel uses an external direct current to drive nickel ions from an anode onto the stainless steel substrate.
- High-Volume Efficiency: It is generally faster to deposit and significantly more cost-effective for large-volume production runs, such as flat sheet metal brackets.
- The Faraday Cage Effect: Electrolytic plating struggles severely with complex geometries. Electrical current will not reach into shielded internal cavities or blind holes, leaving them unplated.
- Current Density Variations: High-current areas—such as sharp CNC corners or sheared edges—experience rapid material build-up known as the “dog-bone effect.” Conversely, low-current flat centers plate much thinner. This uneven build-up easily pushes tightly toleranced parts out of spec.
How Machining and Forming Dictate Nickel Plating Quality?
Shop-floor machining operations directly dictate the final fit and reliability of a plated component. Geometry, tolerance planning, and surface topography all interact with the plating process.
Tolerance Compensation
Engineers often overlook that surface treatments add physical material to the substrate. If an engineering drawing specifies a 15-micron Electroless Nickel Plating (ENP) layer, the CNC machinist must precisely calculate this volumetric build-up.
To maintain final assembly tolerances, shafts must be machined 15 microns undersized per side (30 microns total on the diameter), and bores must be oversized by the exact same amount before the part ever leaves the shop floor.
Thread Clearance
Internal threads are a primary failure point for plated mechanical components. Standard tap sizes will not work; the plating thickness alters the pitch diameter by approximately four times the coating thickness.
Machinists must utilize specialized pre-plate taps—such as 6G or custom H-limit taps—to guarantee the threads will accept standard hardware without galling or binding after the nickel is applied.
Surface Roughness (Ra)
A common manufacturing misconception is that a thick nickel layer will smooth out poor machining marks. In reality, ENP perfectly replicates the underlying topography of the substrate.
If a CNC part leaves the mill with a rough 3.2 Ra (µm) finish due to aggressive tool feeds, the plated part will simply become a shiny, hardened 3.2 Ra surface. Critical sealing surfaces must be ground or polished to their final Ra spec prior to plating. Plating does not hide poor machining; it highlights it.
Sheet Metal Edges
Stamped or laser-cut stainless steel sheet metal requires mechanical edge radiusing before entering the plating line. Sharp, un-deburred edges act as high-current density zones during electrolytic plating, causing severe, brittle material build-up.
Furthermore, the Heat-Affected Zone (HAZ) generated by laser cutting fundamentally alters the local metallurgy. This edge carbonization actively resists chemical activation, leading to localized plating voids and peeling.
Root Causes of Nickel Plating Failures on the Shop Floor
Plating failures are rarely mystery anomalies; they are direct results of process deviation or inadequate surface preparation.

Poor Adhesion
When nickel flakes off a stainless substrate during a thermal shock or cross-hatch adhesion test (ASTM D3359), the root cause rarely lies in the primary nickel bath. It almost universally points to a depleted Wood’s Nickel Strike or inadequate degreasing.
Silicate residues from alkaline cleaners, or synthetic cutting fluids left trapped in blind holes, will instantly destroy the metallurgical bond between the substrate and the nickel.
Edge Burning
“Burning” is a severe defect in electrolytic plating where the nickel coating becomes dark, rough, and highly brittle at the extremities of the part. This occurs when the current density is pushed far too high to accelerate production throughput.
Plating engineers must design custom racking systems equipped with current “robbers” or “thieves”—metal wire additions that deliberately divert excess current away from sharp corners to ensure an even deposition.
Hydrogen Embrittlement
The aggressive acidic activation required to strip the passive layer from stainless steel introduces atomic hydrogen directly into the metal lattice. For high-strength or precipitation-hardened stainless grades (like 17-4 PH or 400 series), this trapped hydrogen can cause sudden, catastrophic cracking under mechanical load.
To mitigate this severe risk, these specific alloys must undergo a strict hydrogen relief baking cycle. Industry standards dictate the parts must be baked at 190°C within exactly four hours of exiting the plating line.
Welded Areas
Welded assemblies are notoriously difficult to plate uniformly due to heavy oxide scale and altered microstructures. The intense heat from TIG or MIG welding creates a localized surface condition that standard acid pickling cannot penetrate.
Chemical activation is insufficient on its own; welded seams must be mechanically abraded, wire-brushed, or bead-blasted prior to the chemical strike to ensure the nickel adheres properly to the joint.
Supplier Audit Checklist: Vetting a Plating Partner
Before awarding a high-volume contract, verify these parameters with your surface finishing supplier:
- “What is your maximum transfer time between the Wood’s strike and the main nickel bath?” (Any duration over 60 seconds is a red flag for repassivation.)
- “How do you handle internal threads?” (They should proactively request your pre-plate tap limits.)
- “What is your baking window for 17-4 PH stainless?” (Failure to strictly enforce the 4-hour rule puts your parts at immediate risk of hydrogen embrittlement.)
Material Selection and the True Cost of Surface Finishing
Material choice dictates whether plating is a practical necessity or an avoidable expense. Different stainless grades shift the cost-to-performance balance.
304 vs. 316 Stainless Steel
Not all stainless steels behave equally on the plating line. Standard 304 stainless is relatively straightforward to activate using standard shop parameters.
However, marine-grade 316 stainless contains molybdenum, making its passive oxide layer significantly more chemically resistant. This demands a more aggressive, tightly monitored Wood’s strike to achieve baseline adhesion, often resulting in higher piece-price quotes.
High-Strength Stainless Steel
Martensitic grades (like 420 or 440C) and hardened alloys introduce high levels of carbon into the metal matrix. When exposed to activation acids, these elements do not dissolve, leaving behind a black, powdery “smut” on the surface.
If this carbon smut is not completely removed via a specialized anodic electro-cleaning step, the nickel layer will deposit directly onto dirt, guaranteeing massive adhesion failure in the field.
Nickel Plating vs. Solid Alloys
Procurement managers must calculate the true break-even point of surface finishing during the Design for Manufacturability (DFM) phase. Is it actually cheaper to machine a standard 304 part and pay for a high-phosphorus ENP treatment to achieve wear resistance?
Once you factor in external logistics, longer lead times, and potential plating scrap rates, machining the part directly from a premium wear-resistant superalloy is often the smarter commercial decision.
Lifecycle Cost
The lowest quote from a plating supplier is frequently the most expensive option over a product’s lifecycle. A poorly controlled, porous 10-micron nickel layer might save pennies on the initial purchase order.
However, trying to cut costs on a sloppy Wood’s strike is a false economy. If that inferior plating flakes off and causes a tightly toleranced stainless steel thread to gall, you may be forced to scrap an entire high-value fluid pump assembly.
When Nickel Plating Is the Wrong Engineering Choice?
Nickel plating is not a universal solution. Depending on the mechanical environment and material interfaces, alternative surface treatments often yield superior performance and lower scrap rates.
Passivation
Do not specify nickel plating if the primary engineering requirement is strict rust prevention. If the stainless steel simply needs to survive a corrosive environment without altering its mechanical properties, standard passivation (ASTM A967) is the correct call.
Using nitric or citric acid baths removes free iron from the surface and artificially thickens the natural chromium oxide layer. This process is highly cost-effective and introduces absolutely zero dimensional changes to tightly toleranced CNC parts.
Hard Chrome
Electroless nickel is hard, but it will fail under extreme sliding wear, abrasive friction, or heavy impact loading (such as hydraulic cylinder shafts). In these environments, the nickel layer will eventually chip or gall.
Industrial Hard Chrome plating is the superior choice for these applications. It provides a significantly lower coefficient of friction and a much higher baseline hardness before any post-plate heat treatments are applied.
Zinc-Nickel
Nickel is highly cathodic. If a nickel-plated stainless steel component is bolted directly to an aluminum housing in a wet environment, it will cause the softer aluminum to rapidly deteriorate through galvanic corrosion.
For mixed-metal aerospace or automotive assemblies, Zinc-Nickel plating is required. It provides sacrificial protection, actively corroding first to protect both the stainless fastener and the surrounding aluminum structure.
PVD Coatings
When dimensional tolerances are restricted to single-digit microns, even a thin ENP layer is out of spec. Physical Vapor Deposition (PVD) coatings, such as Titanium Nitride (TiN), offer extreme surface hardness (over 2000 HV) at a fraction of the thickness.
Applied in a vacuum chamber, PVD typically deposits only 2 to 5 microns of material. It is the industrial standard for high-end injection mold tooling and medical cutting instruments where edge sharpness must be perfectly maintained.
Quick Reference Guide:
- Need pure rust prevention without dimensional shift? Use Passivation.
- Need extreme impact resistance and low friction? Use Hard Chrome.
- Bolting the part directly into aluminum? Use Zinc-Nickel.
- Need a sharp edge with zero tolerance shift? Use PVD.
Mitigating Plating Defects on the Shop Floor
Consistent plating requires strict surface preparation and deliberate design choices. Plating defects are rarely chemical anomalies; they are usually failures in process control or geometry.
Pretreatment Control
The most critical tank in a plating facility is the alkaline degreaser. If a supplier extends the life of their cleaning chemistry to cut operating costs, synthetic cutting fluids and stamping lubricants will survive the wash.
These microscopic oil residues carry over into the acid activation tanks, immediately neutralizing the Wood’s strike. This guarantees mass adhesion failures across the entire production batch.
Blind Holes
Deep blind holes in CNC machined manifolds are a severe liability for wet processing. Chemical solutions become trapped in these cavities, leading to “bleed-out”—a defect where highly corrosive acids leak out days later, destroying the surrounding nickel layer.
If blind holes cannot be designed out during the engineering phase, mandate custom racking orientations and aggressive ultrasonic cleaning to force trapped fluids out of the internal recesses.
Drainage Design
Plating chemistry is expensive, and suppliers penalize designs that cause “drag-out” (scooping up bath fluid and carrying it into the next tank). Deep-drawn sheet metal enclosures often act like buckets on the processing line.
During the Design for Manufacturability (DFM) phase, always integrate strategic weep holes or drainage cutouts into your sheet metal designs. This eliminates chemical cross-contamination, accelerates rinsing times, and actively lowers your piece-price quote.
RoHS and REACH Compliance
Never assume a standard ENP bath is environmentally compliant. Historically, chemical suppliers utilized trace amounts of heavy metals—specifically lead and cadmium—as stabilizers to prevent the bath from degrading.
For global product distribution, mandate written certification that your supplier utilizes modern, heavy-metal-free chemistry. Failing to secure this documentation will result in the entire product assembly failing RoHS and REACH compliance, triggering customs seizures and severe supply chain delays.
Conclusion
Nickel plating stainless steel is not just a finishing step. It is a process that directly affects fit, durability, and long-term performance. When the surface is not prepared correctly, even a good coating will fail in production.
The final result always depends on how well the design, machining, and plating process are controlled together. Small decisions in tolerance, surface finish, or material grade can change both quality and cost.
If you are working on CNC parts, sheet metal components, or stainless steel assemblies that require nickel plating, we can help you review the design before production and reduce plating risks early in the process. Send us your drawings or requirements. We will evaluate feasibility, suggest process options, and provide a clear manufacturing solution for your project.




